This is the 2nd half of a two-part post about MS. In this part, I explore infections likely to cause MS. It took me 3 weeks of research—I hope it helps someone you love. Refer to Part 1 for the basics of MS, its epidemiology suggesting infection, and the lackluster outcomes from current MS treatments.
Recap of the End of Part 1
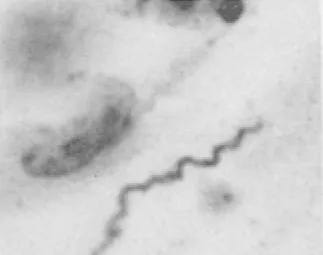
Over the four decades since 1913, when researchers first documented transmission of neurologic disease from MS patients to animals via inoculation, interest in potential infectious causes of MS exploded. Spirochetes, a type of bacteria, were repeatedly found by microscopic examination of both MS patients’ brains and animals into which MS tissue was injected. Although many replicated these findings, often only some animals got sick, whereas some researchers failed to reproduce transmission.
Spirochetes couldn’t be grown alive from MS patients or infected animals for over 40 years. That changed in 1957 when Ichelson grew live spirochetes from MS patients’ spinal f…
Keep reading with a 7-day free trial
Subscribe to ZeroSpin to keep reading this post and get 7 days of free access to the full post archives.